
About
Marjolein van Egmond is professor in Oncology and Inflammation. She is PI at the Department of Molecular Cell Biology & Immunology and staff member at the Department of Surgery. Throughout her career she has been fascinated by the role of antibodies in immunity and the activation of immune cells via antibody receptors (Fc receptors). Initially her research focused on the role of the Fc receptor for IgA (FcαRI) in protective mucosal immunity. Subsequently, her lab discovered that aberrant IgA initiates perpetual neutrophil activation, leading to severe tissue damage in multiple auto-immune diseases and chronic inflammation. Additionally it is now clear that antibodies can be used as therapeutic drugs to engage the immune system to fight cancer. Due to her dual appointment at a research- and clinical department, she is in an excellent position to translate experimental findings into clinical applications. Marjolein van Egmond was awarded a VENI, VIDI and VICI from the Innovational Research Incentives Scheme (Netherlands Organization for Scientific Research, NWO).
Research Line
Monoclonal antibody therapy of cancer (theme Tumorimmunology)
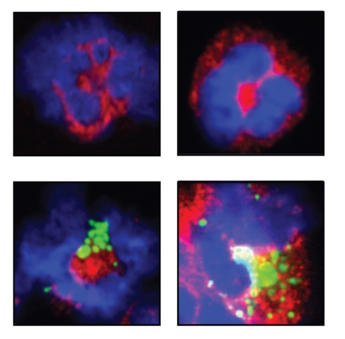
Because neutrophils and macrophages and have potent cytotoxic abilities we are examining whether it is possible to enlist this potential for tumour cell killing. Both myeloid cell types express Fc receptors and are, as such, able to recognize and kill tumour cells in the presence of specific monoclonal antibodies (mAbs) that opsonize tumour cells. We have demonstrated that macrophages are extremely efficient in phagocytosing and digesting circulating cancer cells in the presence of specific IgG mAbs. By contrast, neutrophils induce tumour cell death especially in the presence of monoclonal antibodies of the IgA subclass. We are currently investigating the mechanisms of neutrophil- and macrophage mediated tumour cell killing in more detail, as we anticipate that this knowledge will help us to develop novel (pre-operative) adjuvant anti-cancer therapies.
The role of neutrophil IgA Fc receptor (Fc-alfa-RI) in health and disease. (theme Mucosal Immunology)
IgA is the principal antibody in mucosal areas and plays a key role in mucosal defence. Neutrophils become highly activated when their IgA Fc receptor (FcaRI) is targeted. Since neutrophils are the first cells to arrive at inflammatory (mucosal) sites upon invasion by pathogens, we postulate that activation of neutrophil FcaRI by IgA-coated microorganisms will trigger an essential immune response that will help to clear the infection. However, aberrant IgA deposits are found in several diseases (e.g. IgA-induced blistering diseases and rheumatoid arthritis). We showed that a perpetuating inflammatory loop is initiated when excessive IgA complexes are present, which leads to sever tissue damage. We therefore aim to unravel the role of IgA- induced neutrophil activation in protective and harmful immune responses in more detail.
Key publications
- Breedveld A, van Egmond M. IgA and FcαRI: Pathological Roles and Therapeutic Opportunities. Front Immunol. 2019; 10:553.
- Abis GSA, Stockmann HBAC, Bonjer HJ, van Veenendaal N, van Doorn-Schepens MLM, Budding AE, Wilschut JA, van Egmond M, Oosterling SJ; SELECT trial study group. Randomized clinical trial of selective decontamination of the digestive tract in elective colorectal cancer surgery (SELECT trial). Br J Surg. 2019;106:355-363.
- Grewal S, Korthouwer R, Bögels M, Braster R, Heemskerk N, Budding AE, Pouw SM, van Horssen J, Ankersmit M, Meijerink J, van den Tol P, Oosterling S, Bonjer J, Gül N, van Egmond M. Spillage of bacterial products during colon surgery increases the risk of liver metastases development in a rat colon carcinoma model. Oncoimmunology. 2018;7:e1461302.
- Heineke MH, Ballering AV, Jamin A, Ben Mkaddem S, Monteiro RC, Van Egmond M. New insights in the pathogenesis of immunoglobulin A vasculitis (Henoch-Schönlein purpura). Autoimmun Rev. 2017;16:1246-1253.
- Gül N, Babes L, Siegmund K, Korthouwer R, Bögels M, Braster R, Vidarsson G, ten Hagen TL, Kubes P, van Egmond M. Macrophages eliminate circulating tumor cells after monoclonal antibody therapy. J Clin Invest. 2014;124:812-23.
Group members

Carolien Zeelen, MSc
Research technician
One of the projects I work on is the optimization of circulating tumor cell detection in blood of patients using flow cytometry. The goal of this project is to see if CTCs can be targeted with antibody therapy and therefore prevent metastases. Next to that, I work on the visualization of interaction between immune cells and blood vessels in mouse tumors using fluorescent immunohistochemistry.

Céline Sewnath, MSc
PhD student
The focus of my work is about overcoming the immunosuppressive milieu in order to induce a potent adaptive immune response. For this, I will use new antibody therapies that will target tumor cells but also activate immune cells. Therefore, I will characterize the immune profile in colorectal cancer patients and investigate if these new antibody therapies are able to induce the adaptive immune response and thereby long term anti-tumour immunity. For this project I will use different in vitro assays on primary human cells such as RNA-sequencing data, ADCCs, ADCPs, flow cytometry, cytokine assays (Luminex, ELISA), co-cultures and in vivo work.

Dennis Gout, MSc
PhD student
My main project focuses on the improvement of a neutrophil-targeting, bi-specific antibody therapy for cancers by the addition of an immuno-active payload to the antibody. I’m using these antibody-cytokine fusion proteins (immunocytokines) as a platform to attack the innate immune exclusion in various pre-clinical tumor models from multiple angles. This project involves the design, cloning and production of these immunocytokines as well as the characterization and functional testing of them both in vitro and a pre-clinical tumor model..

Kees Tuk
research technician
Main focus of my work: development of bispecific antibodies for Immunotherapy. All techniques that are necessary for this are used, mainly in vitro techniques. Mostly cell isolation techniques, Cell culture, in vitro cell function assays, ELISA’s. Also: development of in vitro assays to determine cell function of the effect of antibody complexes on cells.

Jasmin I Ersöz
research technician
TBD

Leonie Behrens, MSc
PhD student
My research focuses on the innate immune checkpoint CD47-SIRPα, which limits tumor cell killing by various myeloid cell types. My aim is to determine how SIRPα signaling prevents neutrophil activation, and how blockade of this checkpoint may improve neutrophil-mediated cytotoxicity towards cancer cells. To do so, I perform various in vitro assays to follow the different steps of tumor killing by neutrophils, e.g. formation of an immunological synapse, trogocytosis and tumor cell death.

Mandy Gruijs, MSc
PhD student
My research focuses on antibody therapy in cancer treatment. We have developed a bispecific antibody, which targets both a tumor antigen and neutrophils, as well as macrophages and NK cells. Additionally, I am involved in a clinical trial in which we test an already clinically used antibody in a new regimen. We have expertise in several experimental techniques, including tumor cell killing assays, flow cytometry, microscopy and ELISA.

Myrthe van Delft, PhD
postdoctoral researcher
The focus of my work is studying the role of IgA and FcaR1 in auto-immunity. My aim is to unravel the role of IgA induced (myeloid) cell activation in inflammation and tissue damage in auto-immune diseases (like IgA-induced blistering disease and rheumatoid arthritis). For this I make use of multicolor flow cytometry, cytokine and binding ELISAs, myeloid cell assays and mice models.

Niels Heemskerk, PhD
Senior postdoctoral researcher
My research is focused on the targeting and regulation of selective immune cell trafficking across tumor blood vessels in order to improve immunotherapy of cancer by increasing the entry of antitumor immune cells in tumors.

Paula Winter, BSc
research technician
My research is focused on the IgA Fc receptor (FcaRI) on neutrophils. We look at the difference between this receptor and the IgG Fc receptor (FcyR). And especially the difference in pathway it follows after activation. For this project we use several experimental techniques, including 2d migration, western blot, flow cytometry, chemotaxis and ELISA

Richard van der Mast, BSc
research technician
The projects I am working on are related to antibody therapy in cancer treatment and to the role of the Fc receptor for IgA in mucosal immunity. These projects mainly focus on the role of neutrophils, macrophages and NK cells in the tumor microenvironment or inflammatory (mucosal) sites. I have experience in several techniques, including (live cell) microscopy, tumor cell killing assays, tissue stainings, flow cytometry and ELISA.










